Hot on the heels of the popular term genomics – defined by Wikipedia as “the study of an organism’s entire genome” – comes one for virologists: viromics. There have been a number of articles in recent years on “viromes”, or the total viral genome content of the DNA found in certain biological sampling areas such as entire oceans or just in humans, so one might be forgiven for assuming that viromics was the study of entire viral genomes.
But you might be wrong…for example, Applied Viromics of California have this to say:
“The term “viromics” was introduced on a biochemical pharmacology discussion conference in 2001 (Lotze MT and Kost TA. Cancer Gene Therapy. 2002 Aug; 9(8):692-9). It refers to “the use of viruses and viral gene transfer to explore the complexity arising from the vast array of new targets available from the human and murine genomes”.”
So: just about using viruses to study animals, then? The Katze lab at the University of Washington – who I note have the word in their Web address – focus on:
…the use of genomic and proteomic technologies to study virus-host interactions and the varied strategies used by viruses to evade cellular defense mechanisms. We study a wide range of viral pathogens, including hepatitis C, influenza, Ebola, West Nile, SARS-associated coronavirus, herpes simplex virus, and human and simian immunodeficiency viruses.”
So it’s studying human-virus interactions, then? However, the Marine Microbiology Group at the Universrsity of South Florida has as its viromics interest
“…the interaction of viral genomes with their environments by sequencing several temperate/pseudotemperate marine phage genomes”.
Much more like what I thought it was! However, possibly the best definition I have seen is from a Nature Biotechnology Commentary on “Systems biology and the host response to viral infection“, which pops up the term “Systems virology” as a synonym for viromics. the article states:
“There is increasing evidence from functional genomics experiments that the patterns of cellular response to a variety of viral infections may reflect the pathogenic properties of the viruses. We contend that dissection of the critical, and often subtly different, cellular pathways will eventually unveil opportunities for manipulating the host immune response to fight off viral infection, control pathogenesis or both.”
And in a side box on viromics:
“Innovations in sequencing technologies, particularly the rapid, high-throughput pyrosequencing platforms, continue to transform large-scale biology. … The fields of viral metagenomics and viral diagnostics are therefore poised for rapid expansion.”
Again – unsurprisingly, given that the Katze lab mentioned above is involved – a rather pathocentric approach, but all in all, a nice summary of current thinking.
So it is not the “what” so much as the “what it does” with viruses – at least, with the human virome. Of course, for those of us with other interests, the new viromics can largely be concerned with discovery: there is a largely undescribed universe of viruses out there, in everything from seawater to duck ponds, biofilms to beehives, colons to parrots. Cataloguing some of that diversity can only lead to new insights – which will lead to the kinds of questions that can be answered by “systems virology” approaches.
For instance, in my PhD thesis in 1984, I published an 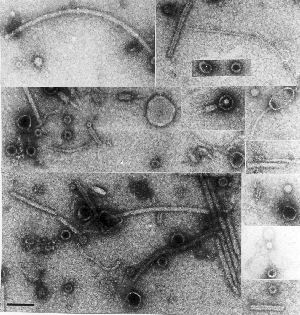 electron micrograph of a collection of unidentified particles found in preparations of known viruses, concentrated and partially purified from plant extracts. there is a veritable zoo of things present, from a variety of fialments – some probably flagellae – to a motley collection of spheres and even some recognisable phages or parts thereof. It has also been stuck on my office wall for the last ten years, since I found it kicking about in a drawer….
electron micrograph of a collection of unidentified particles found in preparations of known viruses, concentrated and partially purified from plant extracts. there is a veritable zoo of things present, from a variety of fialments – some probably flagellae – to a motley collection of spheres and even some recognisable phages or parts thereof. It has also been stuck on my office wall for the last ten years, since I found it kicking about in a drawer….
The point is, we generally find what we are looking for – and presume that what is present in the greatest abundance is just that, and then ignore everything else.
Now we don’t have to do that any more: a metagenomic study of a particle-enriched preparation such as that seen on the right could yield fabulous riches, and unveil hitherto-unsuspected minor populations of plant viruses, unknown fungal and bacterial viruses, and even viruses infecting insects, which may be passively “vectored” by plants.
In a dramatic application of the worth of this sort of discovery, a major metagenomic project involving colony decline disorder of honeybees was described in MicrobiologyBytes recently: this involved sequencing all of the DNA and cDNA from many beehives, and doing a subtractive analysis to determine which possible pathogen was involved.
Other efforts under way in our laboratories in Cape Town include a study of the single-stranded circular DNA virome of grasses in Africa: this has resulted in six publications to date, and over 300 full mastrevirus genomic sequences in less than two years, and has radically altered our perception of both the diversity and the recombination potential of these commercially-important viruses. This phenomenal progress has been enabled by recent technological breakthroughs – in this case, the use of bacteriophage phi29 DNA polymerase for isothermal “rolling circle” genome amplification, and cheap commercial sequencing. This is rapidly diversifying into similar work on Beak and feather disease circoviruses in parrots, and even dsDNA viruses.
We are at the threshold of an era of significant discovery potential in virology, much as happened in the early 1990s with the advent of PCR – only more so. The nice thing is that rapidly-developing sequencing techniques will allow ever-cheaper large-scale sequencing – so that finances will not be the limiting factor they may have been up to now, and developing countries can share in the viromic bounty.
And every now and then, the young people around will tell me what is going on – for which I am grateful.
 West Nile virus
West Nile virus 
