…like a rocket…flashed past; I’m still emotionally in August or so!
I meant to do some more substantive posts instead of only copying Scoop.it Virology News posts here; however, the best-laid plans and such, and I didn’t. I will in 2013, though – and there will be an iBook coming or possibly even two (influenza and PCR), so I will use this forum to announce glad tidings.
Then there’s the ZA Virus [=Zombie Apocalypse, obviously] novel, and Green Vaccines, and…OK, getting ahead of myself here!
Thanks for the support and readership, I hope everyone has a good solstice break!
Best,
Ed
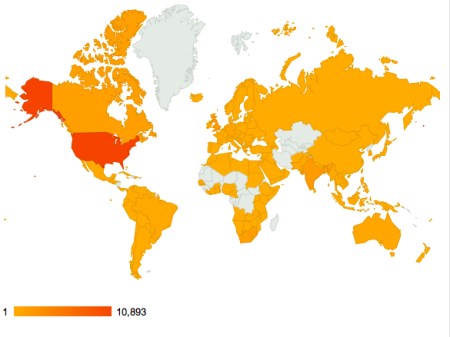
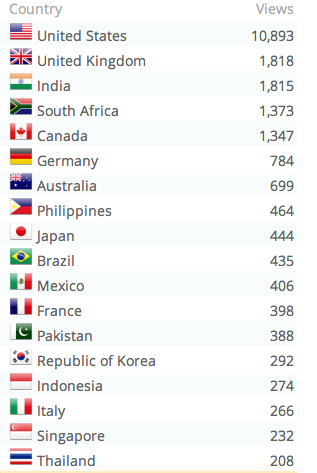
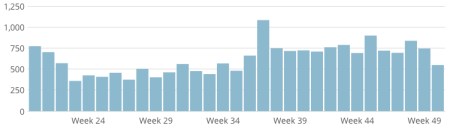
PS: some access stats for 2012 for you. Looks like the only places that DON’T access ViroBlogy are parts of central and west Africa, central Asia and Greenland.