Well, “The Big One” that we have all been waiting for since 1968 – or 1977, if you count the accidental re-release of the original humanised H1N1 as a pandemic – is well and truly here. A nice little animated graphic for depicting how it arose, while a bit simplistic, is available here.
And what have we learned? Has civilisation fallen; have populations been decimated?
Well, quite a lot; no, and no, in answer. Explanations for some of this are contained in a survey just released. Here from the News24 report published today, sourced from SAPA:
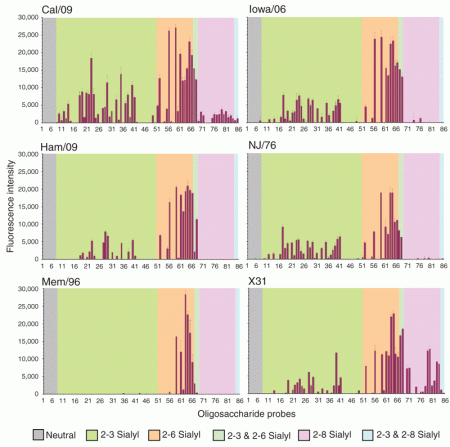
Paris – More than half the fatalities from H1N1 swine flu have been among young adults, according to one of the first surveys to gather mortality data from across the globe for the new A(H1N1) virus.
The analysis of 574 pandemic deaths from 28 countries through mid-July, released this week, also found that being diabetic or obese significantly boosted the risk of dying.
Neither children nor the elderly are as vulnerable as initial reports indicated, found the study, published by Eurosurveillance, the monitoring arm of the European Centre for Disease Prevention and Control.
“Most deaths (51%) occurred in the age group of 20-to-49 year-olds, but there is considerable variation depending on country or continent,” the researchers reported.
Only 12% of those who died were 60 or older.
All of these features – high mortality among young adults and the obese, but not the very young or elderly – are sharply different than for the seasonal flu.
More than 90% of deaths from seasonal flu – which claims 250 000 to 500 000 lives annually according to the WHO – are in people over 65.
By contrast, with the pandemic H1N1, “the elderly seem to be protected from infection to some extent, perhaps due to previous exposure to similar strains”, the study conjectured.
Persons born before 1957, other studies have suggested, were almost certainly exposed to the milder seasonal A(H1N1) viruses that evolved from the terrible pandemic of 1918, which left some 40 million dead.
With the 2009 strain, “when infection does occur, however, the percentage of deaths in elderly cases seems to be higher that in others”.
One common target across both pandemic and season strains is pregnant women, according to the study, led by Philippe Barboza of the French Institute for Public Health Surveillance….
And that’s the sinister part…here in South Africa, of 18 fatalities known to have been associated with pandemic AH1N1 infection, NINE were pregnant women, mostly in the third trimester of pregnancy. In a report published yesterday, SA’s Minister of Health Dr Aaron Motsoaledi said the following:
“We find it very worrying that there is an increasing number of pregnant women who are succumbing to this pandemic,” Motsoaledi said.
“The directive to all health care workers… is to put pregnant women with flu-like symptoms (even if they are mild) on Tamiflu treatment.
“Doctors should not wait for any tests before such treatment is administered.”
Further on in the same report:
On Monday, the National Institute for Communicable Diseases [NICD] also said pregnancy had been identified as a particular risk factor for severe H1N1 flu.
It said that in the second and especially the third trimester, urgent treatment with antiviral drugs should be considered even before any laboratory results were received.
The institute added however that most H1N1 flu cases in South Africa remained mild and “self-limiting”.
Routine H1N1 testing for everyone with flu-like illness was still not recommended.
Nationwide, there had been 5 118 laboratory-confirmed cases of H1N1 flu, it said.
The figure is essentially meaningless, given that most suspected flu cases are not laboratory-diagnosed (it costs R700, or ~US$70, for a single test) and the pandemic flu is pretty much indistinguishable from seasonal, and may in fact have supplanted the normal flu. It certainly has in Australia and Argentina, which remain the two worst-hit southern hemisphere countries, and probably has in South Africa too: the CDC has a very useful map illustrating this, accessible here.
International news, via the CDC site, is the following:
As of August 13, the World Health Organization (WHO) regions have reported over 182,166 laboratory-confirmed cases of 2009 H1N1 influenza virus (2009 H1N1) with 1,799 deaths. The laboratory-confirmed cases represent an underestimation of total cases in the world as many countries now focus surveillance and laboratory testing only in persons with severe illness. The 2009 H1N1 influenza virus continues to be the dominant influenza virus in circulation in the world.
One very important piece of information further down this report is the following:
There have been no significant changes detected in the 2009 H1N1 influenza virus isolated from persons in the Southern Hemisphere as compared to viruses isolated from persons in the Northern Hemisphere.
This is important because the frantic rush to make vaccines to combat the expected northern hemisphere upsurge in infections in their autumn season – October or so – depends upon the virus not having changed much from the seed material which was derived from virus isolated earlier this year. This could negate some of theh fears that the much-anticipated “second wave” of virus infections could be a lot worse than the first.
Good news on the vaccine front – for Australians at least – is that an Australian company, CSL Ltd, has the world’s first data from human trials of a pandemic strain vaccine, and looks set to be able to provide Australia with 21 million doses of vaccine – and 2 million doses of the vaccine at the end of the month.
Other vaccine news is also fairly encouraging, notwithstanding a rather alarming report in New Scientist recently about the new strain growing only half as well in eggs as seasonal flu types: while this remains a worry, newer, faster-growing variants have been derived and distributed – though possibly not in time for a northern hemisphere autumn roll-out.
Mind you, all of this production relies on the well-proven-but-seriously-archaic 1930s technology of growing live virus in hen’s eggs: we are still trapped, in the 21st century, into having to use early 20th century methods to produce vaccines for fast-adapting pathogens. Things ARE changing: various pharma companies are diversifying into mammalian and insect cell culture; people (including us!) are investigating making recombinant subunit vaccines in plants (see here) – and there is at least the tantalising possibility that “universal vaccines” may become available in the not-too-distant future. These will exploit all or part of the highly conserved M2 “ion channel” protein of influenza viruses as recombinant subunit vaccines.
However, all of this is at least six months in the future for conventional vaccines, and many years hence for newer offerings. Meantime – there is disturbing news concerning trans-species transmissions of pandemic AH1N1 viruses.
ProMED Mail (ProMED Digest V2009 #394) reports that “Chile finds H1N1 swine flu in turkeys“:
Chilean health authorities announced on Thursday night [20 Aug 2009] that they had detected and controlled an outbreak of swine flu in 2 turkey farms, according to a communication from the Agricultural and Livestock Service (SAG).
“The presence of an influenza type A virus was detected in 2 farms in the Valparaiso Region, and immediate precautionary measures were adopted to prevent the dissemination of the disease and to protect the population’s health,” said the text.
And again from ProMED on 20th August, quoting The Straits Times and AFP:
A 2nd Australian piggery was placed in quarantine due to swine flu on Wednesday [19 Aug 2009] as the number of human deaths from the virus reached 121.
Authorities ordered a biosecurity lockdown at the piggery in Victoria state amid concerns the virus could mutate and return to humans in a more deadly form.
Another piggery in New South Wales state has been quarantined since late July [2009], although the state government said most of the animals had recovered from the disease.
Victoria Agriculture Minister Joe Helper said tests confirmed the presence of influenza at the piggery after its owners reported earlier this week that the animals were not eating.
‘It is important to stress that this is not a human health issue and that national and international food authorities continue to advise that pork and pork products are safe to eat,’ he said.
Media reports said the pigs were believed to have contracted the virus from workers at the property who were suffering the human form of the disease.
Health experts fear swine flu in humans, which is easily spread but has a relatively low fatality rate, could mutate in other animals and emerge in a more virulent form. [my emphasis]
So: two independent incidents, on different continents, of pandemic AH1N1 viruses getting into different species of farmed livestock – and luckily controlled.
What would have happened if domestic fowl and/or pigs had been infected in places like Vietnam, Thailand, Indonesia, Turkey and Egypt – where highly pathogenic avian H5N1 influenza viruses appear to be endemic, and not well controlled? Given the complex origins of the current pandemic virus – from several swine, avian and human viruses – it could be a recipe for disaster, on a scale even greater than the 1918 pandemic.
The REAL Big One. Let’s all help get a vaccine, people!!