See on Scoop.it – Virology News

Date: Tue 31 Jul 2012
Source: Time Healthland, Associated Press report [edited]
http://healthland.time.com/2012/07/30/6-more-ugandans-admitted-with-possible-ebola/
More Ugandans Admitted with Possible Ebola
——————————————
A total of 6 more patients suspected to have Ebola have been admitted to the hospital days after investigators confirmed an outbreak of the highly infectious disease in a remote corner of western Uganda, a health official said on Monday [30 Jul 2012]. Stephen Byaruhanga, health secretary of the affected Kibaale district, said possible cases of Ebola, at 1st concentrated in a single village, are now being reported in more villages. “It’s no longer just one village. There are many villages affected,” Byaruhanga said. In a national address on Monday, Uganda’s President advised against unnecessary contact among people, saying suspected cases of Ebola should be reported immediately to health officials.
Officials from Uganda’s Ministry of Health and the World Health Organization announced on Saturday [28 Jul 2012] that the deadly Ebola virus killed 14 Ugandans this month, ending weeks of speculation about the cause of a strange illness that had some people fleeing their homes in the absence of reliable answers. If the 6 new cases are confirmed as Ebola fever, it would bring to 26 the number of Ugandans infected with Ebola [virus].
This is the 4th occurrence of Ebola in Uganda since 2000, when the disease killed 224 people and left hundreds more traumatized in norther Uganda. At least 42 people were killed in another outbreak in 2007, and there was a lone Ebola case in 2011. Investigators took nearly a month to confirm Ebola’s presence in Uganda this year. In Kibaale, a district with 600 000 residents, some villagers started abandoning their homes to escape what they thought was an illness caused by bad luck. One family lost 9 members, and a clinical officer and her 4-month-old baby died from Ebola, Byaruhanga said.
D.K. Lwamafa, of Uganda’s Ministry of Health, told reporters on Saturday that one Ebola patient from Kibaale had been referred to the national hospital in the capital but had then died in Kibaale.
The confirmation of Ebola’s presence in the area has spread anxiety among sick villagers, who are refusing to go the hospital for fear they don’t have Ebola and will contract it there. All suspected Ebola patients have been isolated at one hospital where patients admitted with other illnesses fled after Ebola was announced. Only the hospital’s maternity ward still has patients, officials said, highlighting the deadly reputation of Ebola in a country where the authorities do not always respond quickly and effectively to emergencies and disasters. Barnabas Tinkasimire, a lawmaker from the area, said that some nurses refused to look after Ebola patients after one clinical officer died and another was taken ill.
“They are saying, ‘We can’t remain here if there is no sufficient allowance’,” Tinkasimire said of medical officers handling Ebola cases. The lawmaker said the government’s response so far has been poor and that it would have been worse without the technical support of organizations such as the World Health Organization and the U.S. Centers for Disease Control and Prevention (CDC). “It took long for the government to respond, and up to now many people don’t know how to guard against Ebola. We need sensitization,” he said.
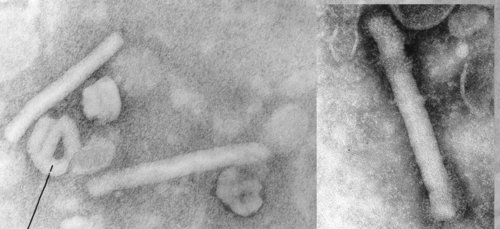
Ebola, which manifests itself as a hemorrhagic fever [But not in this outbreak – Mod.CP], is highly infectious and kills quickly. It was 1st reported in 1976 in Congo and is named for the river where it was recognized. A CDC factsheet on Ebola says the disease is “characterized by fever, headache, joint and muscle aches, sore throat, and weakness, followed by diarrhea, vomiting, and stomach pain. A rash, red eyes, hiccups and internal and external bleeding may be seen in some patients.”
Scientists don’t know the natural reservoir of the virus, but they suspect the 1st victim in an Ebola outbreak gets infected through contact with an infected animal. The virus can be transmitted through direct contact with the blood or secretions of an infected person, or objects that have been contaminated with infected secretions. During communal funerals, for example, when the bereaved come into contact with an Ebola victim, the virus can be contracted, health officials said.
—
Communicated by:
ProMED-mail from HealthMap alerts
See on www.promedmail.org